TBX6 compound inheritance leads to congenital vertebral malformations in humans and mice
- 新闻
Congenital vertebral malformations (CVM) are associated with human TBX6 compound inheritance which combines a rare null allele and a common hypomorphic allele at the TBX6 locus. Our previous in vitro evidence suggested that this compound inheritance resulted in a TBX6 gene dosage of less than haploinsufficiency (i.e. < 50%) as a potential mechanism of 6 TBX6-associated CVM. To further investigate this pathogenetic model, we ascertained and collected 108 Chinese CVM cases and found ten (9.3%) of them carried TBX6 null mutations in combination with common hypomorphic variants at the second TBX6 allele. For in vivo functional verification and genetic analysis of TBX6 compound inheritance, we generated both null and hypomorphic mutations in mouse Tbx6 using the CRISPR-Cas9 method. These Tbx6 mutants are not identical to the patient variants at the DNA sequence level, but instead functionally mimic disease-associated TBX6 variants. Intriguingly, as anticipated by the compound inheritance model, a high penetrance of CVM phenotype was only observed in the mice with combined null and hypomorphic alleles of Tbx6. These findings are consistent with our experimental observations in humans and supported the dosage effect of TBX6 in CVM etiology. In conclusion, our findings in newly collected human CVM subjects and Tbx6 mouse models consistently support the contention that TBX6 compound inheritance causes CVM, potentially via a gene dosage-dependent mechanism. Furthermore, mouse Tbx6 mutants mimicking human CVM-associated variants will be useful models for further mechanistic investigations of CVM pathogenesis in the cases associated with TBX6.
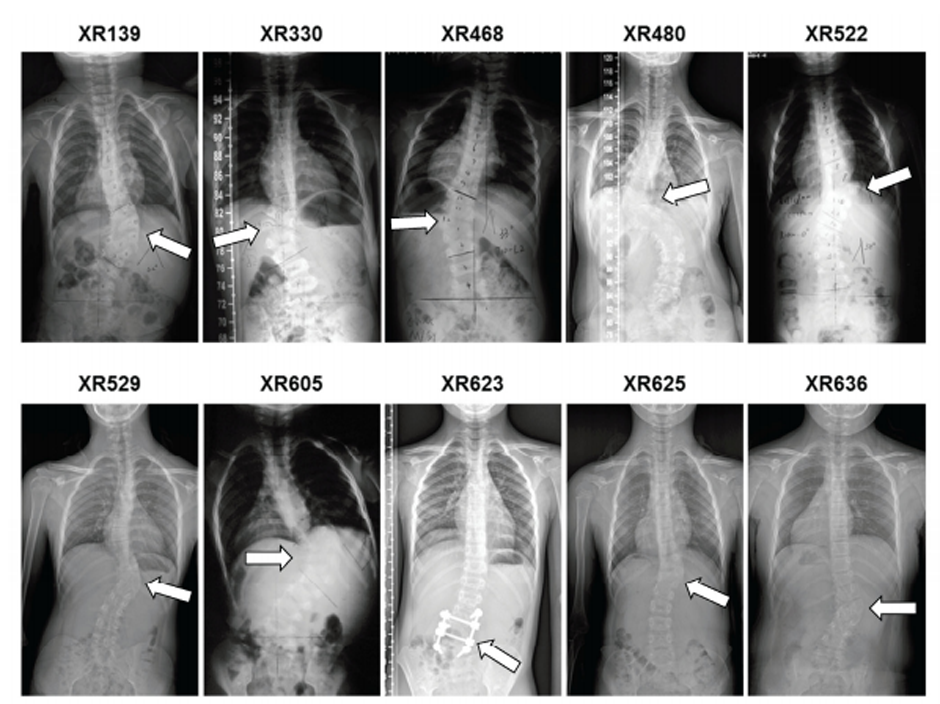 Figure 1. CVM phenotypes of the human subjects associated with TBX6 compound inheritance
Figure 1. CVM phenotypes of the human subjects associated with TBX6 compound inheritance
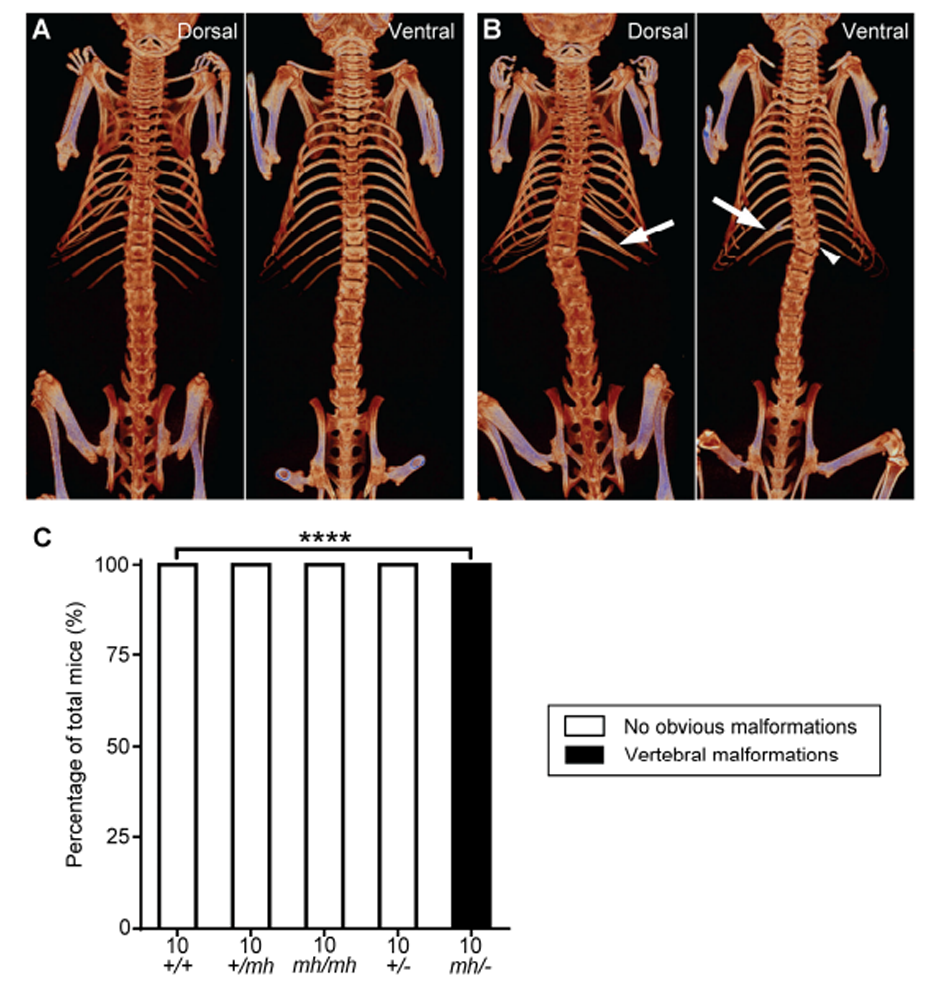
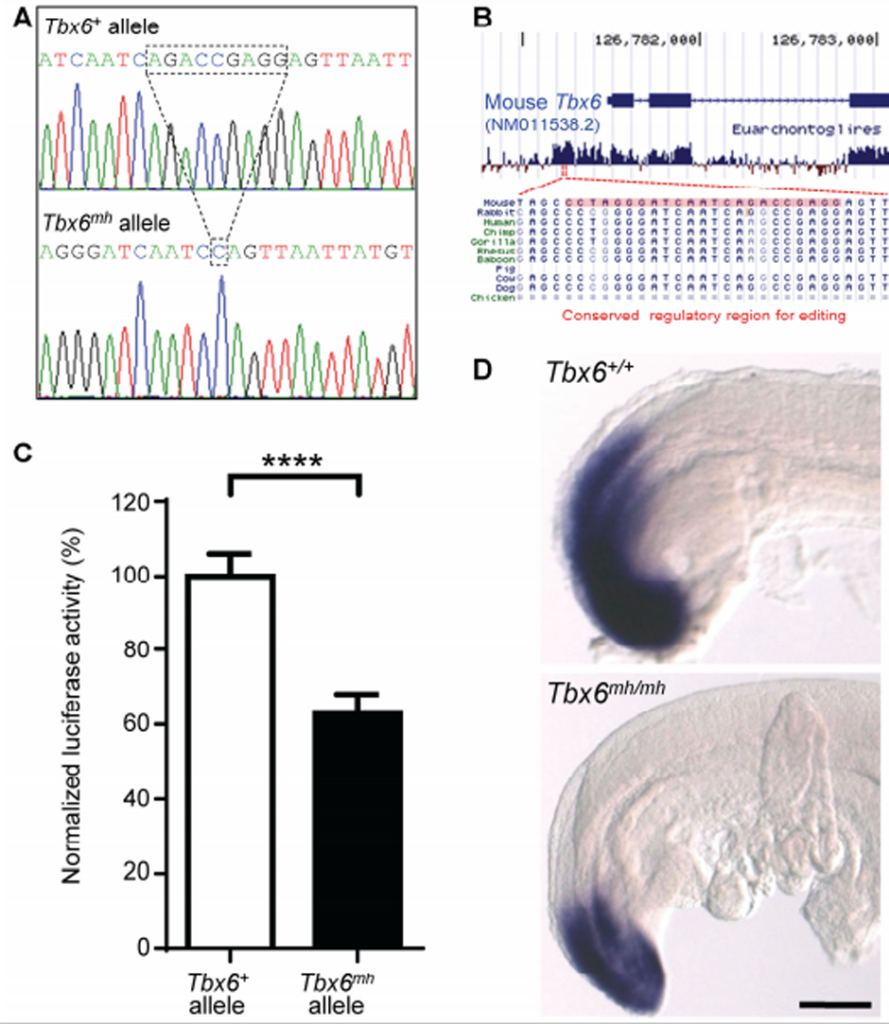 Figure 2. Generation of Tbx6 mild-hypomorphic (mh) mutation in mice
Figure 2. Generation of Tbx6 mild-hypomorphic (mh) mutation in mice
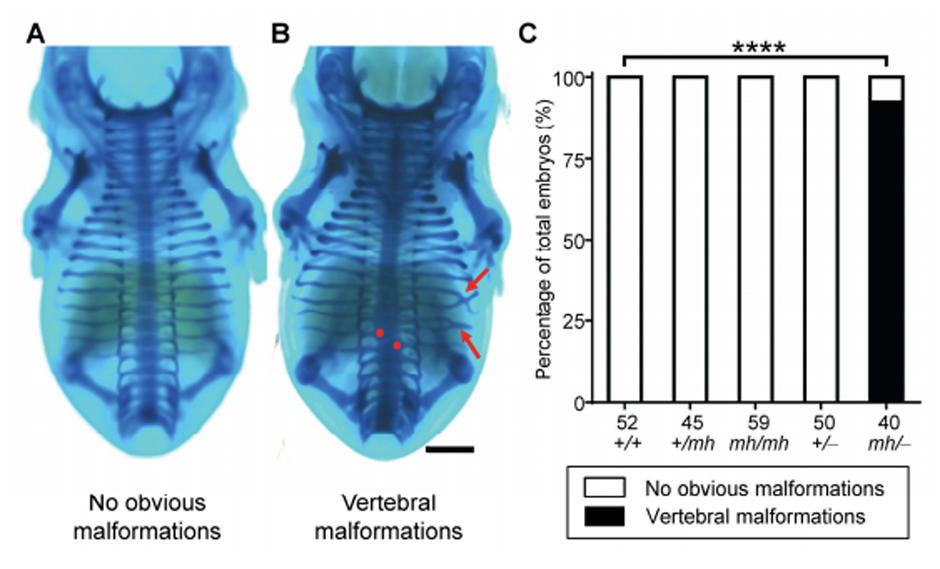 Figure 3. In vivo assays using mouse embryos support the compound inheritance model, 2 consisting of a Tbx6 null allele (‒) and a mild-hypomorphic allele (mh), for the dosage-dependent 3 manifestation of CVM.
Figure 3. In vivo assays using mouse embryos support the compound inheritance model, 2 consisting of a Tbx6 null allele (‒) and a mild-hypomorphic allele (mh), for the dosage-dependent 3 manifestation of CVM.